Healthcare providers are under constant pressure to reduce costs and increase efficiency to improve standards of care while also preventing cross-contamination. Maintaining both HCP and patient safety depends on following aseptic procedures in the operating room, and even then, infections in the operating room still happen.
Healthcare-associated infection (HAI) is a major health and economic issue, and surgical site infection (SSI) is a major contributor. The rate of SSI is influenced by a number of different factors. Microorganisms, such as bacteria, viruses and fungi can potentially contaminate the OR environment through HCPs and staff clothing or surgical equipment or instruments. OR staff behaviour is also an influence; breaks in sterile technique, such as improper hand hygiene, failure to maintain a sterile field, or a glove breach, can increase the risk that pathogens be introduced into the surgical site.
Despite protocols, infections remain a major concern in the surgical environment. SSIs can lead to a requirement for more care, meaning prolonged hospital stays, staff time, additional medication, devices, energy use and possibly further surgical interventions, which all increase healthcare costs and mortality while also being less sustainable.
Infection incidence
It is estimated that one out of every 20 patients undergoing surgery will develop a surgical site infection (SSI)1 and staff exposure rate to patient’s blood during surgery is as high as 10.4 per 100 procedures2. It’s important to ensure patient safety in the operating room (OR) and to protect healthcare professionals from infection from bloodborne illnesses.
In Europe, surgical site infections affect half a million people every year and represent an additional cost of 19 billion euros3.
Infection exposure
Healthcare professionals are frequently exposed to pathogens. Needlestick injuries, of which there are an estimated one million in Europe each year4, put them at risk of contracting bloodborne illnesses, including hepatitis B, hepatitis C and HIV from patients’ blood.
The burden of surgical site infections
Surgical site infections are costly to treat; they also cause pain and trauma to patients. An SSI could, for example, mean that a patient goes on to develop a deep tissue infection, which results in the patient having to remain hospitalised for a longer period of time, requiring more staff time, medication, devices, energy use and potentially further surgical interventions, and all of these have an environmental impact and could all have been prevented.
SSIs are caused by bacteria that get in through incisions made during surgery. The microorganisms enter by one of the following:
- From the skin of patients
- From the surgeons and other operating room personnel
- From the air
- From contaminated surfaces or instruments used during surgery5
Preventing SSI isn’t easy. For example, it’s estimated that about one-third of the population naturally carries the bacteria, Staphylococcus aureus, on the skin and in the nostrils6. Some strains of this bacteria (e.g. methicillin-resistant Staphylococcus aureus, MRSA) are multidrug-resistant organisms. Both staphylococci and enterococci bacteria can survive for days to months after drying on commonly used hospital fabrics and plastics7.
The air is also a source of potential contamination. Humans shed thousands of skin scales every minute8, each potentially carrying bacteria (80% of bacteria come from the air9).
Gloving to prevent infection
Preventing infection and cross-contamination in the OR involves considering the air, patients, staff, equipment and surfaces. We collaborate with healthcare professionals to find the most efficient ways to prevent infection and cross-contamination.
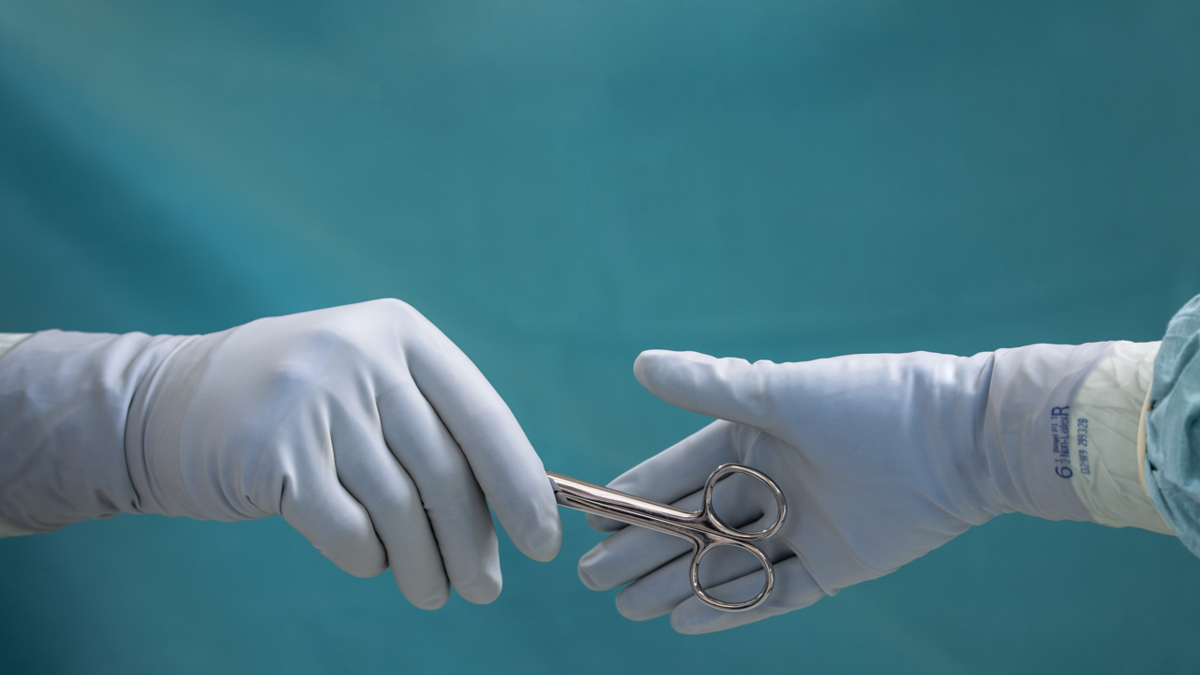
Surgical gloves
Single gloving:
Surgical gloves help reduce the risk of infection for both healthcare professionals and patients10, 11 – and are one of many factors that work together to prevent contamination in the OR10. But if a glove is punctured by a needle, the protection is lost and the hole may not be visible. There is an increased risk of SSI when gloves are punctured11.
Glove powder may be used to make it easier to put on and take off surgical gloves. But it can trigger reduced resistance to infection, bacterial environmental contamination, foreign body reaction, delayed wound healing, adhesion formation and granuloma formation12, 13. All of these potential consequences can increase the risk of SSI14 .
Double gloving with puncture indication:
Double gloving with a puncture indication system – wearing both overgloves and undergloves – enables the healthcare professional to see a glove puncture as soon as it occurs. This means they can quickly change gloves and continue the procedure without compromising protection from cross-infection15, 16.
In a study of gloves used in surgical procedures, the rate of detection of perforations during surgery was 90.2% with the indicator gloves sub-group, compared to 23% and 36.0% with the combination (two regular) gloves and single gloves sub-groups, respectively15.
The practice of double gloving is proven to reduce the risk of needlestick and other sharps injuries and exposure to bloodborne infections. A 2014 Cochrane Review concludes that double gloving reduces the risk of an inner glove perforation by 71%, compared to single gloving16.
Leading authorities now recommend double gloving for invasive surgeries:
- Centers for Disease Control and Prevention (CDC)17
- National Association of Theatre Nurses (NATN)18
- Association of periOperative Registered Nurses (AORN)19
- American College of Surgeons (ACS)20
- Royal College of Surgeons of England18
- Australian College of Operating Room Nurses18
Preventing infection with staff clothing
Staff clothing plays a key role in infection prevention – through protecting patients and OR staff from contamination and supporting clean air in the OR.
Multi-patient (reusable) scrub suits
Traditional, textile scrub suits are washed between surgeries and reused. However, the use of multi-patient (reusable) textiles raises concerns about the effective cleanliness of the laundered clothing and potential risks from lint production, human health and safety concerns, and environmental impact.
Cleanliness of multi-patient scrub suits
Stains, wear and tear, and invisible dangers like bacteria can compromise protection and quality of performance.
Firstly, can effective laundry facilities be guaranteed, whether the facility is in-house or delivered by a third party? Often, laundry facilities harbour pathogens even in clean environments, leading to recontamination after disinfection. In one study, an epidemiologist determined that the laundry was a potential unrecognised vector of hospital-acquired infections21.
The difficulty of effectively removing recalcitrant bacteria, such as clostridium was demonstrated in a large academic university hospital in the United States. It was determined that high levels of CDIFF existed both in their dirty and clean areas of the laundry as well as on delivery truck handles transporting processed textiles back to the facility22.
Increased risk of airborne particles due to laundry comes from lint production. Airborne particles increase the risk of SSI, and lint contamination of surgical wounds contributes to other complications, such as thrombogenesis (blood clots), infections, amplified inflammation, poor wound healing, granulomas, adhesions and capsule formation.
Health and human safety of multi-patient scrub suits
Occupational health is a critical component of sustainability, and by extension, a key part of ensuring occupational health and human safety is reducing the risk of exposure to contaminated material for all staff. With multi-patient (reusable) textiles, laundering reusables poses occupational health and safety risks23, for wearers, patients and laundry staff alike. Hospital laundry workers, for example, handle reusables repeatedly and are at risk of exposure to contaminated material and infection in hospital laundry facilities.
Environmental impact of multi-patient (reusable) scrub suits
Multi-patient (reusable) scrub suits also have a clear environmental impact. The environmental footprint of laundering reusables includes water consumption24, AMR pathogens entering wastewater25, and biocides and detergents entering wastewater. In addition, laundry facilities require a great deal of hot water, which consumes a lot of energy. In fact, the laundry consumes approximately 50-75% of the total hot water generated in a hospital26. Finally, laundry transport and delivery functions also contribute to CO2 emissions.
Single-patient (single-use) scrub suits
Single-patient (single-use) scrub suits contribute to hospital infection control measures. Polypropylene scrub suits in particular are associated with significantly lower air and wound bacterial contamination than conventional cotton shirts and trousers27.
Wearing single-patient scrub suits ensures that the wearer has a new, quality-controlled product every time. There is no risk of damage or contamination from previous use.
Clean air suits
Ultra clean air in the OR is achieved by a combination of laminar air flow, the number of door openings, the number of people in the OR and clean air suits. It’s particularly important in orthopaedic, implant and paediatric interventions.
Clean air suits are especially designed to reduce contamination from airborne microorganisms by staff. They have a better protective effect than a multi-patient (reusable) system made of cotton/ polyester, giving high resistance towards bacterial penetration28 even when ordinary ventilation is used in the OR29.
Preventing contamination from the patient’s skin
Surgical drapes and whole body washing can prevent microorganisms from the patient’s skin from contaminating the surgical wound and causing SSIs.

Surgical drapes
Surgical drapes prevent the transfer of microorganisms from the patient’s skin in surgical procedures. Impermeable materials control infection, while effective fluid control allows for a drier working area. The most effective drapes have fewer parts and are easy to use – to reduce the risk of contamination.
Washing the skin
Whole body washing reduces the skin bacterial load and ultimately decreases the risk of SSI. There are a number of ways to clean the skin:
- Soap and water: the typical standard patient-cleansing procedure. But this simply spreads the organisms, releasing the bacteria over the surface of the skin from the naturally heavily colonized areas30.
- Povidone iodine: preoperative body washing with povidone iodine provides some protection. But surgery-associated products like blood, bus and fat make it less effective31.
- Chlorhexidine digluconate solution: in several studies, a chlorhexidine digluconate solution has been found to prevent healthcare-acquired conditions in intensive care settings32. Recent surgical protocols recommend patients shower twice with chlorhexidine digluconate solution before surgery, waiting at least one minute before rinsing it off33. It’s also less likely to cause skin reactions than alternatives34.
Reducing potential infection vectors with surgical procedure trays
In addition to increasing efficiency and convenience, surgical procedure trays like ProcedurePak reduce the potential for introducing additional sources of microorganisms in the OR35. All the components, surgical instruments, and procedure-specific surgical drapes and gowns required to perform a surgery are contained within a single package. The pack forms the basis for not only less preparation time, less opening and set-up time³⁸ but also to reduce the risk of contamination in the OR due to less packages to open³⁹.
All-in-one and single use
ProcedurePak is designed to include every single-patient component required for a single surgical procedure within a single package, meaning there are fewer packages to open, reduced waste in the OR36, and less risk for contamination37.
The all-in-one convenience of ProcedurePak also allows for a faster OR set-up, which contributes to an overall reduction in procedure duration, which has also been shown to reduce the risk of SSI.
